3.1 The Cell Membrane
Learning Objectives
By the end of this section, you will be able to:
- Describe the molecular components that make up the cell membrane
- Relate structures of the cell membrane to its functions
- Describe how molecules cross the cell membrane based on their properties and concentration gradients
- Compare and contrast different types of passive transport with active transport, providing examples of each
Despite differences in structure and function, all living cells in multicellular organisms have a surrounding cell membrane. Just as the outer layer of your skin separates your body from its environment, the cell membrane (also known as the plasma membrane) separates the inner contents of a cell from its exterior environment. This cell membrane provides a protective barrier around the cell and regulates which materials can pass in or out.
Structure and Composition of the Cell Membrane
The cell membrane is an extremely pliable structure composed primarily of two layers of phospholipids (a “bilayer”). Cholesterol and various proteins are also embedded within the membrane giving the membrane a variety of functions described below.
A single phospholipid molecule has a phosphate group on one end, called the “head,” and two side-by-side chains of fatty acids that make up the lipid “tails” (Figure 3.1.1). The lipid tails of one layer face the lipid tails of the other layer, meeting at the interface of the two layers. The phospholipid heads face outward, one layer exposed to the interior of the cell and one layer exposed to the exterior (Figure 3.1.1).
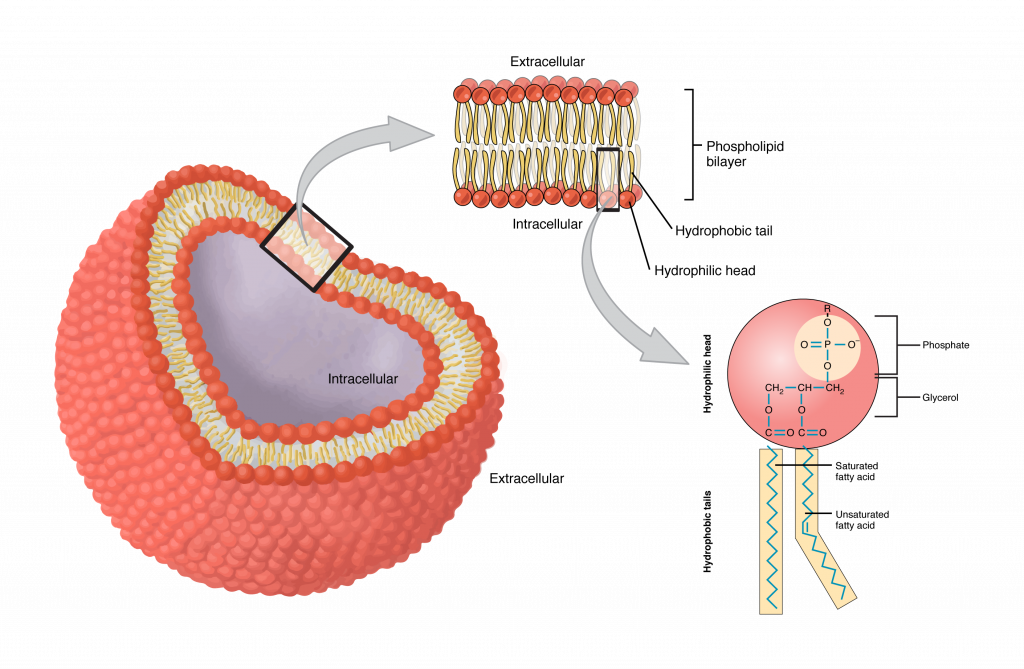
The phosphate group is negatively charged, making the head polar and hydrophilic—or “water loving.” A hydrophilic molecule (or region of a molecule) is one that is attracted to water. The phosphate heads are thus attracted to the water molecules of both the extracellular and intracellular environments. The lipid tails, on the other hand, are uncharged, or nonpolar, and are hydrophobic—or “water fearing.” A hydrophobic molecule (or region of a molecule) repels and is repelled by water. Phospholipids are thus amphipathic molecules. An amphipathic molecule is one that contains both a hydrophilic and a hydrophobic region. In fact, soap works to remove oil and grease stains because it has amphipathic properties. The hydrophilic portion can dissolve in the wash water while the hydrophobic portion can trap grease in stains that then can be washed away. A similar process occurs in your digestive system when bile salts (made from cholesterol, phospholipids and salt) help to break up ingested lipids.
Since the phosphate groups are polar and hydrophilic, they are attracted to water in the intracellular fluid. Intracellular fluid (ICF) is the fluid interior of the cell. The phosphate groups are also attracted to the extracellular fluid. Extracellular fluid (ECF) is the fluid environment outside the enclosure of the cell membrane (see above Figure). Since the lipid tails are hydrophobic, they meet in the inner region of the membrane, excluding watery intracellular and extracellular fluid from this space. In addition to phospholipids and cholesterol, the cell membrane has many proteins detailed in the next section.
Membrane Proteins
The lipid bilayer forms the basis of the cell membrane, but it is peppered throughout with various proteins. Two different types of proteins that are commonly associated with the cell membrane are the integral protein and peripheral protein (Figure 3.1.2). As its name suggests, an integral protein is a protein that is embedded in the membrane. Many different types of integral proteins exist, each with different functions. For example, an integral protein that extends an opening through the membrane for ions to enter or exit the cell is known as a channel protein. Peripheral proteins are typically found on the inner or outer surface of the lipid bilayer but can also be attached to the internal or external surface of an integral protein.
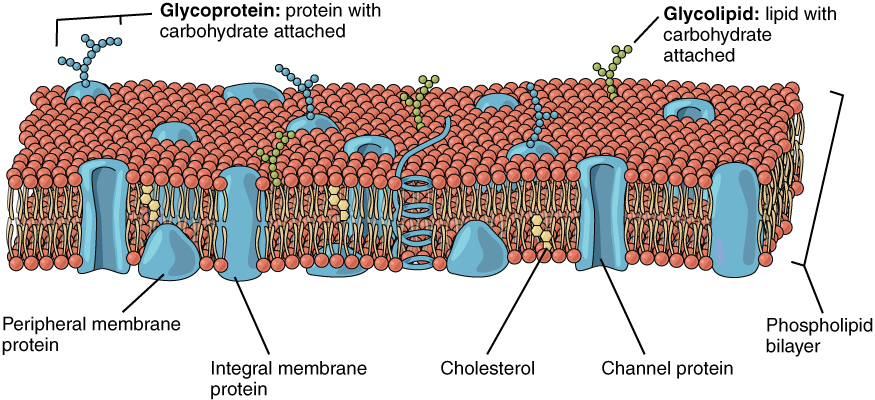
Some integral proteins serve as cell recognition or surface identity proteins, which mark a cell’s identity so that it can be recognized by other cells. Some integral proteins act as enzymes, or in cell adhesion, between neighboring cells. A receptor is a type of recognition protein that can selectively bind a specific molecule outside the cell, and this binding induces a chemical reaction within the cell. Some integral proteins serve dual roles as both a receptor and an ion channel. One example of a receptor-channel interaction is the receptors on nerve cells that bind neurotransmitters, such as dopamine. When a dopamine molecule binds to a dopamine receptor protein, a channel within the transmembrane protein opens to allow certain ions to flow into the cell. Peripheral proteins are often associated with integral proteins along the inner cell membrane where they play a role in cell signaling or anchoring to internal cellular components (ie: cytoskeleton discussed later).
Some integral membrane proteins are glycoproteins. A glycoprotein is a protein that has carbohydrate molecules attached, which extend into the extracellular environment. The attached carbohydrate tags on glycoproteins aid in cell recognition. The carbohydrates that extend from membrane proteins and even from some membrane lipids collectively form the glycocalyx. The glycocalyx is a fuzzy-appearing coating around the cell formed from glycoproteins and other carbohydrates attached to the cell membrane. The glycocalyx can have various roles. For example, it may have molecules that allow the cell to bind to another cell, it may contain receptors for hormones, or it might have enzymes to break down nutrients. The glycocalyces found in a person’s body are products of that person’s genetic makeup. They give each of the individual’s trillions of cells the “identity” of belonging in the person’s body. This identity is the primary way that a person’s immune defense cells “know” not to attack the person’s own body cells, but it also is the reason organs donated by another person might be rejected.
Transport Across the Cell Membrane
One of the great wonders of the cell membrane is its ability to regulate the concentration of substances inside the cell. These substances include ions such as Ca++, Na+, K+, and Cl–, nutrients including sugars, fatty acids, and amino acids, and waste products, particularly carbon dioxide (CO2), which must leave the cell.
The membrane’s lipid bilayer structure provides the first level of control. The phospholipids are tightly packed together, and the membrane has a hydrophobic interior. This structure causes the membrane to be selectively permeable. A membrane that has selective permeability allows only substances meeting certain criteria to pass through it unaided. In the case of the cell membrane, only relatively small, nonpolar materials can move through the lipid bilayer (remember, the lipid tails of the membrane are nonpolar). Some examples of these are other lipids, oxygen and carbon dioxide gases, and alcohol. However, water-soluble materials—like glucose, amino acids, and electrolytes—need some assistance to cross the membrane because they are repelled by the hydrophobic tails of the phospholipid bilayer. All substances that move through the membrane do so by one of two general methods, which are categorized based on whether or not energy is required. Passive transport is the movement of substances across the membrane without the expenditure of cellular energy. In contrast, active transport is the movement of substances across the membrane using energy from adenosine triphosphate (ATP).
Passive Transport
In order to understand how substances move passively across a cell membrane, it is necessary to understand concentration gradients and diffusion. A concentration gradient is the difference in concentration of a substance across a space. Molecules (or ions) will spread/diffuse from where they are more concentrated to where they are less concentrated until they are equally distributed in that space. (When molecules move in this way, they are said to move down their concentration gradient, from high concentration to low concentration.) Diffusion is the movement of particles from an area of higher concentration to an area of lower concentration. A couple of common examples will help to illustrate this concept. Imagine being inside a closed room. If a bottle of perfume were sprayed, the scent molecules would naturally diffuse from the spot where they left the bottle to all corners of the room, and this diffusion would go on until the molecules were equally distributed in the room. Another example is a spoonful of sugar placed in a cup of tea. Eventually the sugar will diffuse throughout the tea until no concentration gradient remains. In both cases, if the room is warmer or the tea hotter, diffusion occurs even faster as the molecules are bumping into each other and spreading out faster than at cooler temperatures.
External Website

Visit this link to see diffusion and how it is propelled by the kinetic energy of molecules in solution. How does temperature affect diffusion rate, and why?
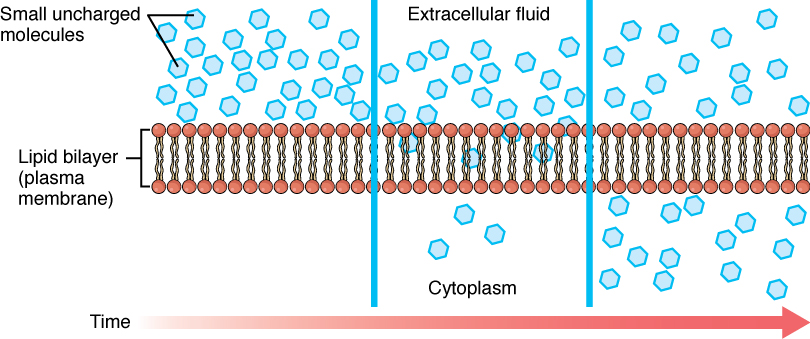
Whenever a substance exists in greater concentration on one side of a semipermeable membrane, such as cell membranes, any substance that can move down its concentration gradient across the membrane will do so. If the substances can move across the cell membrane without the cell expending energy, the movement of molecules is called passive transport. Consider substances that can easily diffuse through the lipid bilayer of the cell membrane, such as the gases oxygen (O2) and carbon dioxide (CO2). These small, fat soluble gasses and other small lipid soluble molecules can dissolve in the membrane and enter or exit the cell following their concentration gradient. This mechanism of molecules moving across a cell membrane from the side where they are more concentrated to the side where they are less concentrated is a form of passive transport called simple diffusion. O2 generally diffuses into cells because it is more concentrated outside of them, and CO2 typically diffuses out of cells because it is more concentrated inside of them.
Before moving on, it is important to realize that the concentration gradients for oxygen and carbon dioxide will always exist across a living cell and never reach equal distribution. This is because cells rapidly use up oxygen during metabolism and so, there is typically a lower concentration of O2 inside the cell than outside. As a result, oxygen will diffuse from outside the cell directly through the lipid bilayer of the membrane and into the cytoplasm within the cell. On the other hand, because cells produce CO2 as a byproduct of metabolism, CO2 concentrations rise within the cytoplasm; therefore, CO2 will move from the cell through the lipid bilayer and into the extracellular fluid, where its concentration is lower. (Figure 3.1.3).
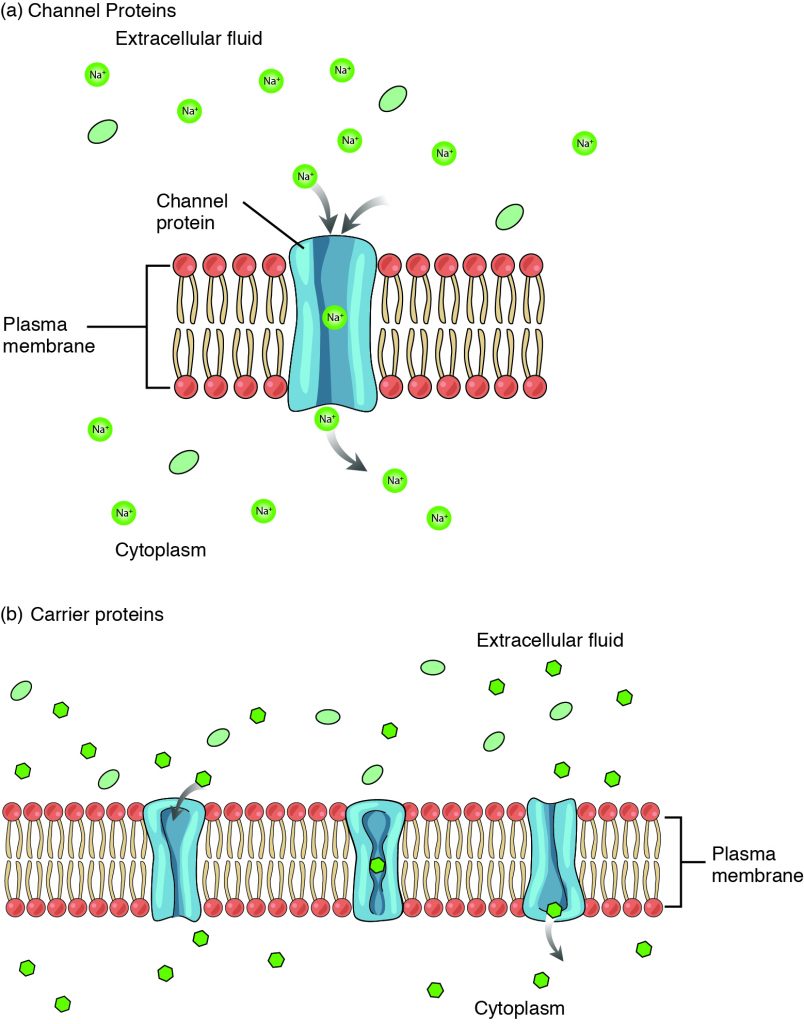
Large polar or ionic molecules, which are hydrophilic, cannot easily cross the phospholipid bilayer. Charged atoms or molecules of any size cannot cross the cell membrane via simple diffusion as the charges are repelled by the hydrophobic tails in the interior of the phospholipid bilayer. Solutes dissolved in water on either side of the cell membrane will tend to diffuse down their concentration gradients, but because most substances cannot pass freely through the lipid bilayer of the cell membrane, their movement is restricted to protein channels and specialized transport mechanisms in the membrane. Facilitated diffusion is the diffusion process used for those substances that cannot cross the lipid bilayer due to their size, charge, and/or polarity but do so down their concentration gradients (Figure 3.1.4). As an example, even though sodium ions (Na+) are highly concentrated outside of cells, these electrolytes are charged and cannot pass through the nonpolar lipid bilayer of the membrane. Their diffusion is facilitated by membrane proteins that form sodium channels (or “pores”), so that Na+ ions can move down their concentration gradient from outside the cells to inside the cells. A common example of facilitated diffusion using a carrier protein is the movement of glucose into the cell, where it is used to make ATP. Although glucose can be more concentrated outside of a cell, it cannot cross the lipid bilayer via simple diffusion because it is both large and polar, and therefore, repelled by the phospholipid membrane. To resolve this, a specialized carrier protein called the glucose transporter will transfer glucose molecules into the cell to facilitate its inward diffusion. The difference between a channel and a carrier is that the carrier usually changes shape during the diffusion process, while the channel does not. There are many other solutes that must undergo facilitated diffusion to move into a cell, such as amino acids, or to move out of a cell, such as wastes.
Osmosis
A specialized example of facilitated transport is water moving across the cell membrane of all cells, through protein channels known as aquaporins. Osmosis is the diffusion of water through a semipermeable membrane from where there is more relative water to where there is less relative water (down its water concentration gradient) (Figure 3.1.5).
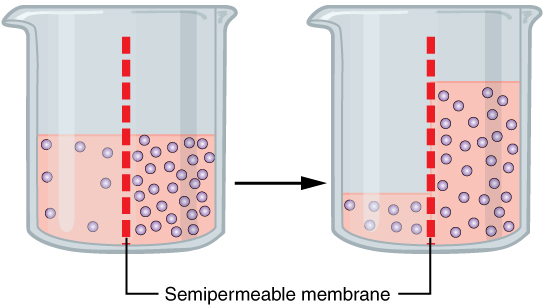
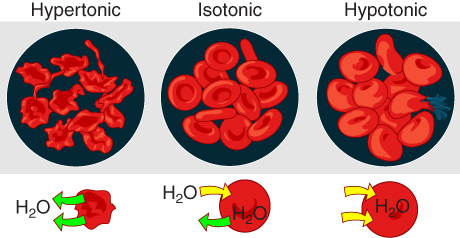
On their own, cells cannot regulate the movement of water molecules across their membrane, so it is important that cells are exposed to an environment in which the concentration of solutes outside of the cells (in the extracellular fluid) is equal to the concentration of solutes inside the cells (in the cytoplasm). Two solutions that have the same concentration of solutes are said to be isotonic (equal tension). When cells and their extracellular environments are isotonic, the concentration of water molecules is the same outside and inside the cells, and the cells maintain their normal shape (and function).
Osmosis occurs when there is an imbalance of solutes outside of a cell versus inside the cell. A solution that has a higher concentration of solutes than another solution is said to be hypertonic, and water molecules tend to diffuse into a hypertonic solution (Figure 3.1.6). Cells in a hypertonic solution will shrivel as water leaves the cell via osmosis. In contrast, a solution that has a lower concentration of solutes than another solution is said to be hypotonic, and water molecules tend to diffuse out of a hypotonic solution. Cells in a hypotonic solution will take on too much water and swell, with the risk of eventually bursting. A critical aspect of homeostasis in living things is to create an internal environment in which all of the body’s cells are in an isotonic solution. Various organ systems, particularly the kidneys, work to maintain this homeostasis.
Active Transport
For all of the transport methods described above, the cell expends no energy. Membrane proteins that aid in the passive transport of substances do so without the use of ATP. During primary active transport, ATP is required to move a substance across a membrane, with the help of membrane protein, and against its concentration gradient.
One of the most common types of active transport involves proteins that serve as pumps. The word “pump” probably conjures up thoughts of using energy to pump up the tire of a bicycle or a basketball. Similarly, energy from ATP is required for these membrane proteins to transport substances—molecules or ions—across the membrane, against their concentration gradients (from an area of low concentration to an area of high concentration).
The sodium-potassium pump, which is also called Na+/K+ ATPase, transports sodium out of a cell while moving potassium into the cell. The Na+/K+ pump is an important ion pump found in the membranes of all cells. The activity of these pumps in nerve cells is so great that it accounts for the majority of their ATP usage.
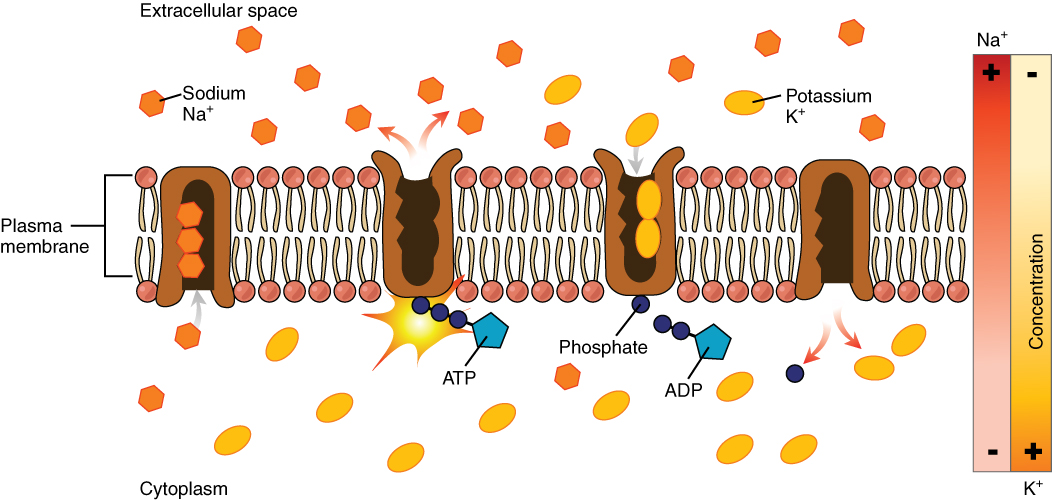
Active transport pumps can also work together with other active or passive transport systems to move substances across the membrane. For example, the sodium-potassium pump maintains a high concentration of sodium ions outside of the cell. Therefore, if the cell needs sodium ions, all it has to do is open a passive sodium channel, as the concentration gradient of the sodium ions will drive them to diffuse into the cell. In this way, the action of an active transport pump (the sodium-potassium pump) powers the passive transport of sodium ions by creating a concentration gradient. When active transport powers the transport of another substance in this way, it is called secondary active transport.
Symporters are secondary active transporters that move two substances in the same direction. For example, the sodium-glucose symporter uses sodium ions to “pull” glucose molecules into the cell. Since cells store glucose for energy, glucose is typically at a higher concentration inside of the cell than outside; however, due to the action of the sodium-potassium pump, sodium ions will easily diffuse into the cell when the symporter is opened. The flood of sodium ions through the symporter provides the energy that allows glucose to move through the symporter and into the cell, against its concentration gradient.
Conversely, antiporters are secondary active transport systems that transport substances in opposite directions. For example, the sodium-hydrogen ion antiporter uses the energy from the inward flood of sodium ions to move hydrogen ions (H+) out of the cell. The sodium-hydrogen antiporter is used to maintain the pH of the cell’s interior.
Other Forms of Membrane Transport
Other forms of active transport do not involve membrane carriers. Endocytosis (bringing “into the cell”) is the process of a cell ingesting material by enveloping it in a portion of its cell membrane, and then pinching off that portion of membrane (Figure 3.1.8). Once pinched off, the portion of membrane and its contents becomes an independent, intracellular vesicle. A vesicle is a membranous sac—a spherical and hollow organelle bounded by a lipid bilayer membrane. Endocytosis often brings materials into the cell that must to be broken down or digested. Phagocytosis (“cell eating”) is the endocytosis of large particles. Many immune cells engage in phagocytosis of invading pathogens. Like little Pac-men, their job is to patrol body tissues for unwanted matter, such as invading bacterial cells, phagocytize them, and digest them. In contrast to phagocytosis, pinocytosis (“cell drinking”) brings fluid containing dissolved substances into a cell through membrane vesicles.
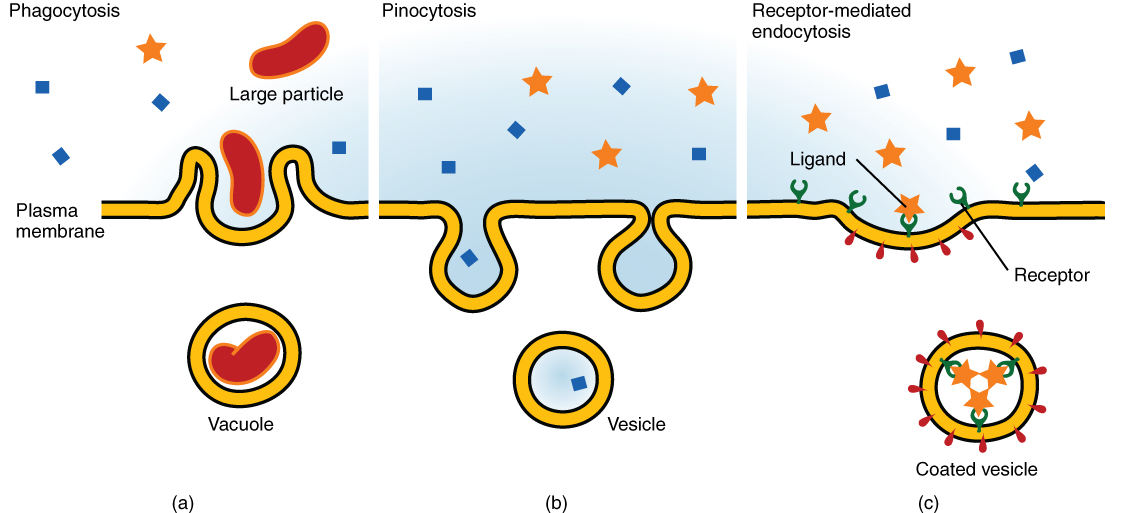
Phagocytosis and pinocytosis take in large portions of extracellular material, and they are typically not highly selective in the substances they bring in. Cells regulate the endocytosis of specific substances via receptor-mediated endocytosis. Receptor-mediated endocytosis is endocytosis by a portion of the cell membrane which contains many receptors that are specific for a certain substance. Once the surface receptors have bound sufficient amounts of the specific substance (the receptor’s ligand), the cell will endocytose the part of the cell membrane containing the receptor-ligand complexes. Iron, a required component of hemoglobin, is endocytosed by red blood cells in this way. Iron is bound to a protein called transferrin in the blood. Specific transferrin receptors on red blood cell surfaces bind the iron-transferrin molecules, and the cell endocytoses the receptor-ligand complexes.
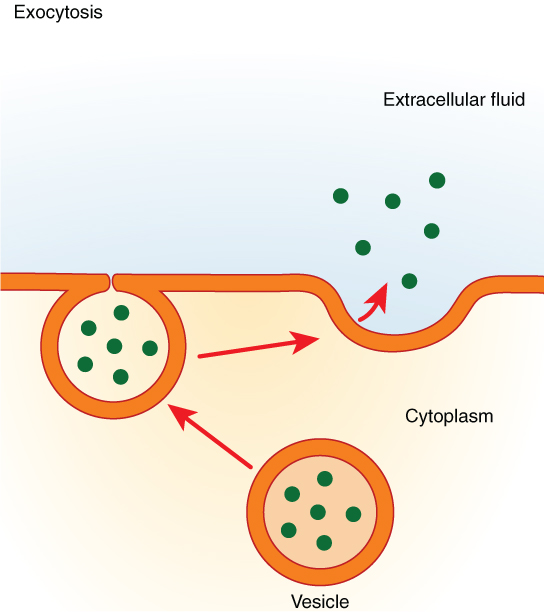
In contrast with endocytosis, exocytosis (taking “out of the cell”) is the process of a cell exporting material using vesicular transport (Figure 3.1.9). Many cells manufacture substances that must be secreted, like a factory manufacturing a product for export. These substances are typically packaged into membrane-bound vesicles within the cell. When the vesicle membrane fuses with the cell membrane, the vesicle releases its contents into the interstitial fluid. The vesicle membrane then becomes part of the cell membrane.
Specific examples of exocytosis include cells of the stomach and pancreas producing and secreting digestive enzymes through exocytosis (Figure 3.1.10) and endocrine cells producing and secreting hormones that are sent throughout the body.
The addition of new membrane to the plasma membrane is usually coupled with endocytosis so that the cell is not constantly enlarging. Through these processes, the cell membrane is constantly renewing and changing as needed by the cell.
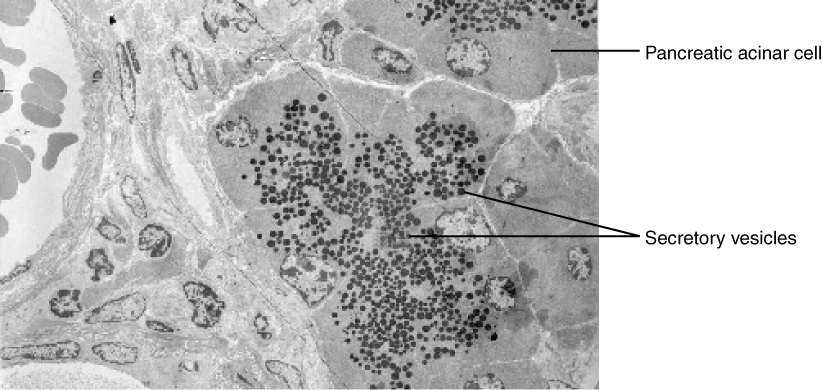
External Website

View the University of Michigan WebScope at http://virtualslides.med.umich.edu/Histology/EMsmallCharts/3%20Image%20Scope%20finals/226%20-%20Pancreas_001.svs/view.apml to explore the tissue sample in greater detail.
Diseases of the Cell: Cystic Fibrosis
Cystic fibrosis (CF) affects approximately 30,000 people in the United States, with about 1,000 new cases reported each year. The genetic disease is most well-known for its damage to the lungs, causing breathing difficulties and chronic lung infections, but it also affects the liver, pancreas, and intestines. Only about 50 years ago, the prognosis for children born with CF was very grim—a life expectancy rarely over 10 years. Today, with advances in medical treatment, many CF patients live into their 30s.
The symptoms of CF result from a malfunctioning membrane ion channel called the Cystic Fibrosis Transmembrane Conductance Regulator, or CFTR. In healthy people, the CFTR protein is an integral membrane protein that transports Cl– ions out of the cell. In a person who has CF, the gene for the CFTR is mutated, thus, the cell manufactures a defective channel protein that typically is not incorporated into the membrane, but is instead degraded by the cell.
The CFTR requires ATP in order to function, making its Cl– transport a form of active transport. This puzzled researchers for a long time because the Cl– ions are actually flowing down their concentration gradient when transported out of cells. Active transport generally pumps ions against their concentration gradient, but the CFTR presents an exception to this rule.
In normal lung tissue, the movement of Cl– out of the cell maintains a Cl–-rich, negatively charged environment immediately outside of the cell. This is particularly important in the epithelial lining of the respiratory system. Respiratory epithelial cells secrete mucus, which serves to trap dust, bacteria, and other debris. A cilium (plural = cilia) is one of the hair-like appendages found on certain cells. Cilia on the epithelial cells move the mucus and its trapped particles up the airways away from the lungs and toward the outside. In order to be effectively moved upward, the mucus cannot be too viscous, rather, it must have a thin, watery consistency. The transport of Cl– and the maintenance of an electronegative environment outside of the cell attracts positive ions such as Na+ to the extracellular space. The accumulation of both Cl– and Na+ ions in the extracellular space creates solute-rich mucus, which has a low concentration of water molecules. As a result, through osmosis, water moves from cells and extracellular matrix into the mucus, “thinning” it out. In a normal respiratory system, this is how the mucus is kept sufficiently watered-down to be propelled out of the respiratory system.
If the CFTR channel is absent, Cl– ions are not transported out of the cell in adequate numbers, thus preventing them from drawing positive ions. The absence of ions in the secreted mucus results in the lack of a normal water concentration gradient. Thus, there is no osmotic pressure pulling water into the mucus. The resulting mucus is thick and sticky, and the ciliated epithelia cannot effectively remove it from the respiratory system. Passageways in the lungs become blocked with mucus, along with the debris it carries. Bacterial infections occur more easily because bacterial cells are not effectively carried away from the lungs.
Chapter Review
The cell membrane provides a barrier around the cell, separating its internal components from the extracellular environment. It is composed of a phospholipid bilayer, with hydrophobic internal lipid “tails” and hydrophilic external phosphate “heads.” Various membrane proteins are scattered throughout the bilayer, both inserted within it and attached to it peripherally. The cell membrane is selectively permeable, allowing only a limited number of materials to diffuse through its lipid bilayer. All materials that cross the membrane do so using passive (non-energy-requiring) or active (energy-requiring) transport processes. During passive transport, materials move by simple diffusion or by facilitated diffusion through the membrane, down their concentration gradient. Water passes through the membrane in a diffusion process called osmosis. During active transport, energy is expended to assist material movement across the membrane in a direction against their concentration gradient. Active transport may take place with the help of protein pumps or through the use of vesicles.
Interactive Link Questions
Visit this link to see diffusion and how it is propelled by the kinetic energy of molecules in solution. How does temperature affect diffusion rate, and why?
Higher temperatures speed up diffusion because molecules have more kinetic energy at higher temperatures.
Review Questions
Critical Thinking Questions
What materials can easily diffuse through the lipid bilayer, and why?
Only materials that are relatively small and nonpolar can easily diffuse through the lipid bilayer. Large particles cannot fit in between the individual phospholipids that are packed together, and polar molecules are repelled by the hydrophobic/nonpolar lipids that line the inside of the bilayer.
Why is receptor-mediated endocytosis said to be more selective than phagocytosis or pinocytosis?
Receptor-mediated endocytosis is more selective because the substances that are brought into the cell are the specific ligands that could bind to the receptors being endocytosed. Phagocytosis or pinocytosis, on the other hand, have no such receptor-ligand specificity, and bring in whatever materials happen to be close to the membrane when it is enveloped.
What do osmosis, diffusion, filtration, and the movement of ions away from like charge all have in common? In what way do they differ?
These four phenomena are similar in the sense that they describe the movement of substances down a particular type of gradient. Osmosis and diffusion involve the movement of water and other substances down their concentration gradients, respectively. Filtration describes the movement of particles down a pressure gradient, and the movement of ions away from a like charge describes their movement down their electrical gradient.
This work, Anatomy & Physiology, is adapted from Anatomy & Physiology by OpenStax, licensed under CC BY. This edition, with revised content and artwork, is licensed under CC BY-SA except where otherwise noted.
Images, from Anatomy & Physiology by OpenStax, are licensed under CC BY except where otherwise noted.
Access the original for free at https://openstax.org/books/anatomy-and-physiology/pages/1-introduction.

