27.2 Development of Sexual Anatomy
Learning Objectives
By the end of this section, you will be able to:
- Explain how bipotential tissues are directed to develop into sex organs
- Name the rudimentary duct systems in the embryo that are precursors to internal sex organs
- Describe the hormonal changes that bring about puberty, and the secondary sex characteristics
Introduction
The development of the sexual systems begins soon after fertilization of the egg, with primordial gonads beginning to develop approximately one month after conception. Sexual system development continues in utero, but there is little change in the system between infancy and puberty.
Development of the Sexual Organs in the Embryo and Fetus
Without chemical prompting, all fertilized eggs would develop a clitoris and vagina. This would be different if an individual was exposed to the cascade of factors initiated by a single gene on the Y chromosome. This is called the SRY (Sex-determining Region of the Y chromosome). Individuals without a Y chromosome also do not have the SRY gene. Without a functional SRY gene, an individual will typically develop a uterus and ovaries.
In all embryos, the same group of cells has the potential to develop into either testes and ovaries; this tissue is considered bipotential. The SRY gene actively recruits other genes that begin to develop the testes, and suppresses other genes that would lead to development of ovaries. As part of this SRY-prompted cascade, germ cells in the bipotential gonads differentiate into spermatogonia. Without SRY, different genes are expressed, oogonia form, and primordial follicles develop in the primitive ovary.
Soon after the formation of the testis, the interstitial (Leydig) cells begin to secrete testosterone. Testosterone can influence tissues that are bipotential. For example, with exposure to testosterone, cells that could become either the glans penis or the glans clitoris form the glans penis. Without testosterone, these same cells differentiate into the clitoris.
Not all tissues in the reproductive tract are bipotential. The internal reproductive structures (for example the uterus, uterine tubes, and part of the vagina; and the epididymis, ductus deferens, and seminal vesicles) form from one of two rudimentary duct systems in the embryo.
Development of the internal sexual organs requires one set of ducts to develop and the other set to degrade. A hormone secreted from sustentacular (Sertoli) cells trigger a degradation of the paramesonephric (Müllerian) duct, and therefore a uterus is unlikely to develop. At the same time, testosterone secretion stimulates growth of the mesonephric (Wolffian) duct, leading to development of the epididymis and vas deferens. Without such sustentacular cell hormone secretion, the paramesonephric duct will now develop; and without testosterone, the mesonephric duct will degrade. Thus, the offspring in this circumstance will likely develop a uterus, and not an epididymis or vas deferens. For more information and a figure of differentiation of the gonads, seek additional content on fetal development.
There are many reasons why sexual anatomy would develop differently than previously described, and it is important to locate intersex anatomy on the spectrum of normal human variation between the binary female and male. In some cases, the receptors that the hormones typically bind to do not develop. For example, in the case of androgen insensitivity, an individual with XY chromosomes, and an SRY gene, will still produce hormones from the sustentacular cells that lead to degradation of the paramesonephric duct – meaning that no uterus can develop. They will also develop testes which will produce testosterone, (androgens) but the cells can not react to the hormones because they lack the receptor to bind the hormone. Therefore, the epididymis and vas deferens are not produced, and the external genitalia develop into a clitoris and vagina. The result is an individual with XY chromosomes, non-descended testes, clitoris and vagina but no uterus.
In contrast to the example above, an intersex condition can result from having hormone secretion beyond what is expected based on the chromosomes. In Congenital Adrenal Hyperplasia, individuals with XX chromosomes have an increase in androgens produced by adrenal glands. The result is a clitoris that is enlarged in size, and at birth may appear similar to a penis. The following image illustrates the spectrum that can exist in clitoral size during adrenal hyperplasia. The increased androgen production in these XX individuals may also lead to increased body hair, receding hair line, deep voice and muscular physique. In an XY individual, a decrease in the expected androgen production can lead to a penis that is much smaller than average, and termed micropenis. This reinforces the notion that external genitalia are developed across a spectrum of size between a clitoris and penis based on the degree of exposure to androgens. This spectrum of normal human variation does not require surgical treatment, only an open mind to the notion of what normal variation might include. Individuals with intersex anatomy have no additional health risks when left to develop on their own, while surgical intervention at a young age includes the risk of surgical complications including nerve damage and infection.
Onset of Puberty
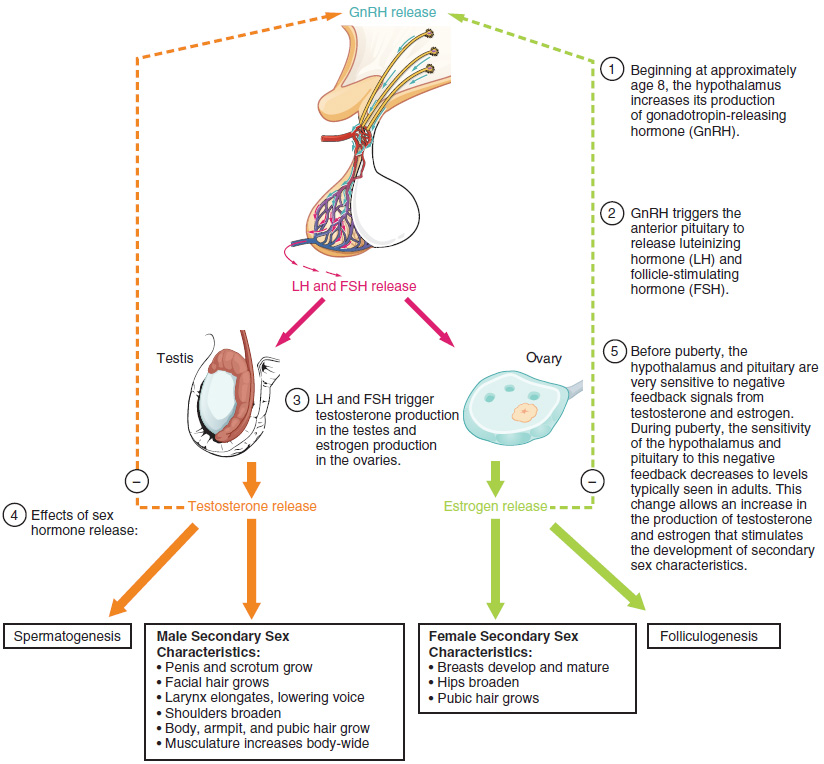
Puberty is the stage of development at which individuals become sexually mature. As shown in Figure 27.2.1, a concerted release of hormones from the hypothalamus (GnRH), the anterior pituitary (LH and FSH), and the gonads (either testosterone or estrogen) is responsible for the maturation of the reproductive systems and the development of secondary sex characteristics, which are physical changes in the body.
The first changes begin around the age of eight or nine when the production of LH becomes detectable. The release of LH occurs primarily at night during sleep and precedes the physical changes of puberty by several years. In pre-pubescent children, the sensitivity of the negative feedback system in the hypothalamus and pituitary is very high. This means that very low concentrations of androgens or estrogens will negatively feed back onto the hypothalamus and pituitary, keeping the production of GnRH, LH, and FSH low.
As an individual approaches puberty, two changes in sensitivity occur. The first is a decrease of sensitivity in the hypothalamus and pituitary to negative feedback, meaning that it takes increasingly larger concentrations of sex steroid hormones to stop the production of LH and FSH. The second change in sensitivity is an increase in sensitivity of the gonads to the FSH and LH signals, meaning the gonads of adults are more responsive to gonadotropins than are the gonads of children. Because of these two changes, the levels of LH and FSH slowly increase and lead to the enlargement and maturation of the gonads, which in turn leads to secretion of higher levels of sex hormones and the initiation of spermatogenesis and folliculogenesis.
In addition to age, multiple factors can affect the age of onset of puberty, including genetics, environment, and psychological stress. One of the more important influences may be nutrition; historical data demonstrate the effect of better and more consistent nutrition on the age of menarche in the United States, which decreased from an average age of approximately 17 years of age in 1860 to the current age of approximately 12.75 years in 1960, as it remains today. Some studies indicate a link between puberty onset and the amount of stored fat in an individual. This effect has been documented in both sexes. Body fat, corresponding with secretion of the hormone leptin by adipose cells, appears to have a strong role in determining menarche. This may reflect to some extent the high metabolic costs of gestation and lactation. In individuals who are lean and highly active, such as gymnasts, there is often a delay in the onset of puberty.
Signs of Puberty
Different sex steroid hormone concentrations also contribute to the development and function of secondary sexual characteristics. Examples of secondary sexual characteristics due to a predominance of testosterone or estrogen are listed in Table 27.1.
An increased production of estrogen at puberty typically leads to the development of breast tissue. This is followed by the growth of axillary and pubic hair. A growth spurt typically starts at approximately age 9 to 11, and may last two years or more. During this time, an individual’s height can increase an average of 3 inches a year. The next step in puberty due to estrogen is menarche, the start of menstruation.
An increased production of testosterone leads to growth of the testes, typically the first physical sign of the beginning of puberty, which is followed by growth and pigmentation of the scrotum and growth of the penis. The next step is the growth of hair, including armpit, pubic, chest, and facial hair. Testosterone stimulates the growth of the larynx and thickening and lengthening of the vocal folds, which causes the voice to drop in pitch. The first fertile ejaculations typically appear at approximately 15 years of age, but this age can vary widely across individuals. The prostate normally doubles in size during puberty. A growth spurt occurs toward the end of puberty, at approximately age 11 to 13, and height can increase as much as 4 inches a year. In some individuals, pubertal development can continue through the early 20s.
Interactive Link Questions
Watch this video to observe ovulation and its initiation in response to the release of FSH and LH from the pituitary gland. What specialized structures help guide the oocyte from the ovary into the uterine tube?
The fimbriae sweep the oocyte into the uterine tube.
Watch this series of videos to look at the movement of the oocyte through the ovary. The cilia in the uterine tube promote movement of the oocyte. What would likely occur if the cilia were paralyzed at the time of ovulation?
The oocyte may not enter the tube and may enter the pelvic cavity.
Review Questions
Critical Thinking Questions
1. Follow the path of ejaculated sperm from the vagina to the oocyte. Include all structures of the female reproductive tract that the sperm must swim through to reach the egg.
2. Identify some differences between meiosis in men and women.
3. Explain the hormonal regulation of the phases of the menstrual cycle.
4. Endometriosis is a disorder in which endometrial cells implant and proliferate outside of the uterus—in the uterine tubes, on the ovaries, or even in the pelvic cavity. Offer a theory as to why endometriosis increases a woman’s risk of infertility.
Glossary
- alveoli
- (of the breast) milk-secreting cells in the mammary gland
- ampulla
- (of the uterine tube) middle portion of the uterine tube in which fertilization often occurs
- antrum
- fluid-filled chamber that characterizes a mature tertiary (antral) follicle
- areola
- highly pigmented, circular area surrounding the raised nipple and containing areolar glands that secrete fluid important for lubrication during suckling
- Bartholin’s glands
- (also, greater vestibular glands) glands that produce a thick mucus that maintains moisture in the vulva area; also referred to as the greater vestibular glands
- body of uterus
- middle section of the uterus
- broad ligament
- wide ligament that supports the uterus by attaching laterally to both sides of the uterus and pelvic wall
- cervix
- elongate inferior end of the uterus where it connects to the vagina
- clitoris
- (also, glans clitoris) nerve-rich area of the vulva that contributes to sexual sensation during intercourse
- corpus albicans
- nonfunctional structure remaining in the ovarian stroma following structural and functional regression of the corpus luteum
- corpus luteum
- transformed follicle after ovulation that secretes progesterone
- endometrium
- inner lining of the uterus, part of which builds up during the secretory phase of the menstrual cycle and then sheds with menses
- fimbriae
- fingerlike projections on the distal uterine tubes
- follicle
- ovarian structure of one oocyte and surrounding granulosa (and later theca) cells
- folliculogenesis
- development of ovarian follicles from primordial to tertiary under the stimulation of gonadotropins
- fundus
- (of the uterus) domed portion of the uterus that is superior to the uterine tubes
- granulosa cells
- supportive cells in the ovarian follicle that produce estrogen
- hymen
- membrane that covers part of the opening of the vagina
- infundibulum
- (of the uterine tube) wide, distal portion of the uterine tube terminating in fimbriae
- isthmus
- narrow, medial portion of the uterine tube that joins the uterus
- labia majora
- hair-covered folds of skin located behind the mons pubis
- labia minora
- thin, pigmented, hairless flaps of skin located medial and deep to the labia majora
- lactiferous ducts
- ducts that connect the mammary glands to the nipple and allow for the transport of milk
- lactiferous sinus
- area of milk collection between alveoli and lactiferous duct
- mammary glands
- glands inside the breast that secrete milk
- menarche
- first menstruation in a pubertal female
- menses
- shedding of the inner portion of the endometrium out though the vagina; also referred to as menstruation
- menses phase
- phase of the menstrual cycle in which the endometrial lining is shed
- menstrual cycle
- approximately 28-day cycle of changes in the uterus consisting of a menses phase, a proliferative phase, and a secretory phase
- mons pubis
- mound of fatty tissue located at the front of the vulva
- myometrium
- smooth muscle layer of uterus that allows for uterine contractions during labor and expulsion of menstrual blood
- oocyte
- cell that results from the division of the oogonium and undergoes meiosis I at the LH surge and meiosis II at fertilization to become a haploid ovum
- oogenesis
- process by which oogonia divide by mitosis to primary oocytes, which undergo meiosis to produce the secondary oocyte and, upon fertilization, the ovum
- oogonia
- ovarian stem cells that undergo mitosis during female fetal development to form primary oocytes
- ovarian cycle
- approximately 28-day cycle of changes in the ovary consisting of a follicular phase and a luteal phase
- ovaries
- female gonads that produce oocytes and sex steroid hormones (notably estrogen and progesterone)
- ovulation
- release of a secondary oocyte and associated granulosa cells from an ovary
- ovum
- haploid female gamete resulting from completion of meiosis II at fertilization
- perimetrium
- outer epithelial layer of uterine wall
- polar body
- smaller cell produced during the process of meiosis in oogenesis
- primary follicles
- ovarian follicles with a primary oocyte and one layer of cuboidal granulosa cells
- primordial follicles
- least developed ovarian follicles that consist of a single oocyte and a single layer of flat (squamous) granulosa cells
- proliferative phase
- phase of the menstrual cycle in which the endometrium proliferates
- rugae
- (of the vagina) folds of skin in the vagina that allow it to stretch during intercourse and childbirth
- secondary follicles
- ovarian follicles with a primary oocyte and multiple layers of granulosa cells
- secretory phase
- phase of the menstrual cycle in which the endometrium secretes a nutrient-rich fluid in preparation for implantation of an embryo
- suspensory ligaments
- bands of connective tissue that suspend the breast onto the chest wall by attachment to the overlying dermis
- tertiary follicles
- (also, antral follicles) ovarian follicles with a primary or secondary oocyte, multiple layers of granulosa cells, and a fully formed antrum
- theca cells
- estrogen-producing cells in a maturing ovarian follicle
- uterine tubes
- (also, fallopian tubes or oviducts) ducts that facilitate transport of an ovulated oocyte to the uterus
- uterus
- muscular hollow organ in which a fertilized egg develops into a fetus
- vagina
- tunnel-like organ that provides access to the uterus for the insertion of semen and from the uterus for the birth of a baby
- vulva
- external female genitalia
Solutions
Answers for Critical Thinking Questions
- The sperm must swim upward in the vagina, through the cervix, and then through the body of the uterus to one or the other of the two uterine tubes. Fertilization generally occurs in the uterine tube.
- Meiosis in the man results in four viable haploid sperm, whereas meiosis in the woman results in a secondary oocyte and, upon completion following fertilization by a sperm, one viable haploid ovum with abundant cytoplasm and up to three polar bodies with little cytoplasm that are destined to die.
- As a result of the degradation of the corpus luteum, a decline in progesterone concentrations triggers the shedding of the endometrial lining, marking the menses phase of the menstrual cycle. Low progesterone levels also reduce the negative feedback that had been occurring at the hypothalamus and pituitary, and result in the release of GnRH and, subsequently, FSH and LH. FSH stimulates tertiary follicles to grow and granulosa and theca cells begin to produce increased amounts of estrogen. High estrogen concentrations stimulate the endometrial lining to rebuild, marking the proliferative phase of the menstrual cycle. The high estrogen concentrations will eventually lead to a decrease in FSH because of negative feedback, resulting in atresia of all but one of the developing tertiary follicles. The switch to positive feedback that occurs with elevated estrogen production from the dominant follicle stimulates the LH surge that will trigger ovulation. The luteinization of the granulosa cells of the collapsed follicle forms the progesterone-producing corpus luteum. Progesterone from the corpus luteum causes the endometrium to prepare for implantation, in part by secreting nutrient-rich fluid. This marks the secretory phase of the menstrual cycle. Finally, in a non-fertile cycle, the corpus luteum will degrade and menses will occur.
- Endometrial tissue proliferating outside of the endometrium—for example, in the uterine tubes, on the ovaries, or within the pelvic cavity—could block the passage of sperm, ovulated oocytes, or a zygote, thus reducing fertility.
This work, Anatomy & Physiology, is adapted from Anatomy & Physiology by OpenStax, licensed under CC BY. This edition, with revised content and artwork, is licensed under CC BY-SA except where otherwise noted.
Images, from Anatomy & Physiology by OpenStax, are licensed under CC BY except where otherwise noted.
Access the original for free at https://openstax.org/books/anatomy-and-physiology/pages/1-introduction.

